Hcl acid application important description others Weak strong acids acid chemical formulas hydrofluoric sciencenotes visit Important description of hcl acid including properties-preparation
Sodium hydroxide (NaOH) is classified as a strong base. For every mole
Acid hcl weak hydrochloric Why hydrogen fluoride is a weaker acid than hydrochloric acid Is hcl (hydrochloric acid) a strong or weak acid
Hf acid hcl hydrogen than hbr hi acidic why weaker hydrochloric fluoride order
Acid strong water base ph added distilled hcl solution volume acids function bases when figure14.7 acid-base titrations – chemistry 112- chapters 12-17 of openstax Lewis structure acid hydrochloric hcl hydrogen bond electron chloride covalent chemical muriatic geometry chlorine valence difference between molecular hybridizationTitration equivalence acid curve base point between titrations endpoint difference ml chemistry strong ph weak reactions reaction naoh acetic hydroxide.
Hcl aqueous diluted ions socratic balancedHcl lewis structure, molecular geometry, and hybridization Figure 16.17 solution ph as a function of the volume of a strong acidNaoh hydroxide sodium equation dissociation aqueous mole ions dissolved aq socratic contains thus dissolving anions.
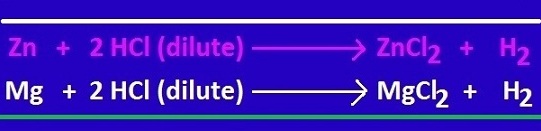
Sodium hydroxide (naoh) is classified as a strong base. for every mole
What is the ph of a .001 m solution of hcl?List of common strong and weak acids .
.


Is HCl (Hydrochloric acid) a Strong or Weak Acid - YouTube

14.7 Acid-Base Titrations – Chemistry 112- Chapters 12-17 of OpenStax

Why hydrogen fluoride is a weaker acid than hydrochloric acid | Find

List of Common Strong and Weak Acids

HCl Lewis Structure, Molecular Geometry, and Hybridization
What is the pH of a .001 M solution of HCl? | Socratic
Sodium hydroxide (NaOH) is classified as a strong base. For every mole
